Where NanoPak C All-Carbon Capillary HPLC Columns Fit in Bottom-Up Proteomics
- Apr 21
- 4 min read
Most bottom-up proteomics methods employ silica-based C18 for peptide mapping and protein digestion analysis. For applications that require stronger retention of very hydrophilic analytes or seek selectivity beyond normal reversed-phase behavior, All-Carbon porous graphitic carbon (PGC) could be a useful addition to proteomics HPLC tools.
This blog explains how NanoPak-C All-Carbon capillary HPLC columns fit into this area. It summarizes our white paper on a preliminary capillary LC–MS comparison using low femtomole BSA tryptic digest. This comparison, performed independently by a collaborator, assessed All Carbon against commercially available PGC and C18 reference capillary columns.
Why Compare All Carbon Media in Proteomics?
PGC is not simply a “stronger C18.” Its retention mechanism combines hydrophobic, polar, and planar interactions. This provides much stronger retention for polar peptides, glycopeptides, glycans, PTM-rich peptides, and metabolites than conventional silica-based reversed-phase columns. For more details on PGC retention, see our separate blog on comparing NanoPak C All Carbon to other commercial PGC-based materials.
We put three types of spherical reverse-phase columns to the test:
C18 (with 5 μm and 3 μm particles)
NanoPak-C All Carbon (with 5 μm and 3.5 μm particles)
Commercial PGC (with 5 μm particles)
All media were packed into 75 μm by 250 mm capillary columns. Each was run under the same conditions, allowing us to directly compare their performance in retention and separation.
What the Chromatograms Showed
The study indicated that PGC-type materials, including All-Carbon, retain digested peptides better than C18 in the BSA digest. The PGC-type columns needed a 5–55% acetonitrile gradient. C18 columns showed strong peptide release in a lower 5–35% acetonitrile range.
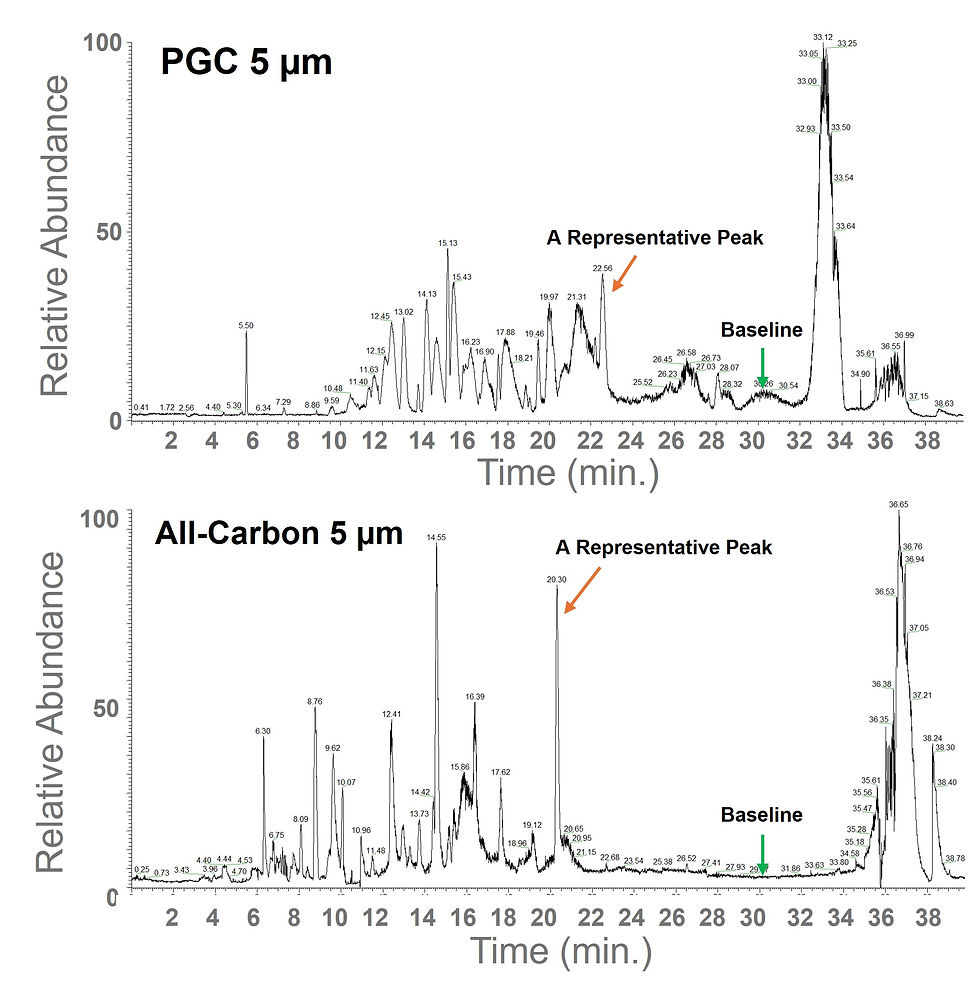
The chromatograms indicated that C18 still yields a denser, more familiar peptide chromatogram profile for a conventional protein digest (see Figures 1a and d). C18 chromatograms show more closely packed, highly resolved peptide peaks across the main elution window. PGC type traces appear more selective, more retentive, and less peak dense overall.
This does not make All-Carbon media less useful. It just shows that all-carbon media should be seen as a complement to C18, not a complete replacement for every proteomics separation.
NanoPak C All Carbon vs Commercial PGC
The more informative comparison was between All Carbon vs. commercially available PGC, since both use graphitic carbon materials tested side by side.
All-Carbon 5 μm (Figure 1c) gave slightly sharper, more efficient peptide peaks than the commercial PGC (Figure 1b) of the same particle size, resulting in better peak separation for more accurate analysis.
All-Carbon 3.5 μm (Figure 1d) improved efficiency and peak separation compared with All-Carbon 5 μm, providing better chromatographic performance for proteomics.
Faster Conditioning. With All Carbon, you don’t have to run as many conditioning injections before the column delivers steady retention and good peak shapes. In our hands, these columns settle in with fewer runs and are much more consistent from one batch to the next. That’s a big plus for proteomics labs—especially when every sample counts and instrument time is at a premium.
A noticeably cleaner baseline. Our test showed that All-Carbon columns provided a cleaner baseline than the commercial PGC columns. In other words, there was less extraneous chemical “background” in our results, making it easier to spot the signals we were actually looking for during our nanoLC–MS tests.
Potential proteomics applications. All Carbon media's unique retention and selectivity could help separate polar compounds that might otherwise overlap or pass through too quickly on a standard C18 column. All-Carbon capillary columns could be useful for separating very hydrophilic peptides, glycopeptides, released glycans, PTM-rich
peptides, or metabolites.
Looking ahead, the All Carbon microbead synthesis technology can be adapted to produce a whole range of carbon-based phases —from those designed for strong polar selectivity to future versions that could approach C18-style peptide mapping.
Read the White Paper
The full chromatographic comparison data, base peak chromatograms for all five columns, experimental conditions, and detailed discussion of retention, efficiency, conditioning, and baseline behavior are available in our white paper:
For more information on our Pre-packed NanoPak-C All-Carbon capillary HPLC columns,
visit our product page.
Standard internal diameter (ID) sizes 75, 100, 200, 500, and 1000 µm
Lengths 5, 10, 15, and 25 cm
Custom ID and lengths available upon request.
The material of capillary columns is a fused silica capillary tube
Compatible with the easy-to-use analytical column hardware (tubings and nuts)
For more information on our NanoPak-C All-Carbon suite of product or to request samples, please email us at inquiry@millennialscientific.com, call us at 855 388 2800 or fill in our online form.
References
K.L. Sanders, J.L. Edwards, Nano-Liquid Chromatography-Mass Spectrometry And Recent Applications In Omics Investigations, Analytical Methods 12(36) (2020) 4404-4417.
West, C., et al. (2010). "Porous Graphitic Carbon: A Cersatile Stationary Phase for Liquid Chromatography." Journal of Chromatography A 1217(19): 3201-3216.
Parente, M. J. and B. Sitharaman (2023). "Synthesis and Characterization of Carbon Microbeads." ACS omega 8(37): 34034-34043.


