Simplifying RNA QC: Can Carbon-Based Digital Liquid Chromatography Help?
- 6 hours ago
- 6 min read
RNA's Therapeutic Promise Meets a Complex QC Reality. Ribonucleic acid (RNA)- based genetic

medicines have unrivaled potential to serve as a platform therapy capable of treating several unmet medical needs, including cancer, genetic diseases, and chronic diseases associated with autoimmune, metabolic, cardiovascular, liver, or lung dysfunction (among others).
Current RNA manufacturing includes mRNA, saRNA, transRNA, circRNA, and related modalities. As these therapeutics move through development and manufacturing, they have created a new analytical challenge for quality control (QC) laboratories. QC teams must evaluate a broad set of critical quality attributes tied to identity, purity, strength, and safety, often with a patchwork of orthogonal methods and instruments. That fragmentation adds instrument footprint, changeover time, and scheduling complexity to an already demanding release testing environment.
Critical Quality Attributes (CQAs) of RNA-lipid nanoparticles (LNPs) currently require weeks of manual assays and a sprawling suite of 10+ disconnected, bulky instruments (HPLC, CE, qPCR, SEC, ELISA). Regulators expect us to demonstrate Safety, Identity, Strength, Purity, Quality (SISPQ) for every lot, as outlined in ICH Q8.
Several of those attributes currently use different liquid chromatography methods (ion-pair reverse-phase, anion-exchange, or size-exclusion). Each LC method, dependent on different physical separation mechanisms based on the RNA’s charge, Size, or Hydrophobicity, requires a dedicated setup.
Electrochemically modulated liquid chromatography (EMLC, or digital liquid chromatography takes a different direction. By applying a potential to an electrically conductive stationary phase, it becomes possible to tune retention in real time, potentially allowing one all-carbon column to interrogate multiple RNA properties that would otherwise require several separate LC methods.
RNA QC is method-intensive because different analytes and impurities exhibit distinct physical properties. Reverse-phase methods are useful to tease out hydrophobic differences, anion-exchange methods probe charge-related behavior, and size-exclusion methods are used to distinguish larger species, such as aggregates, from monomeric material.
Why is RNA QC with digital liquid chromatography interesting? Electrochemically modulated liquid chromatography (EMLC) is an offshoot of high- performance liquid chromatography (HPLC) [1]. EMLC, as a separation technique, merges HPLC and electrochemistry. It uses an electrically conductive stationary phase—most commonly porous graphitic carbon —which also serves as the working electrode in an electrochemical cell.
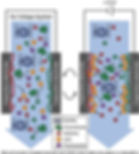
This approach enables manipulation of the interfacial properties of the conductive stationary phase (e.g., donor-acceptor strength and the structure of the electrical double layer) by applying fixed or scanned electrical potentials (Eapp) with a potentiostat. These changes, in turn, affect analyte retention and can even alter the elution order of certain analytes. EMLC is thus distinct from all other forms of liquid chromatography in that the stationary phase may be altered prior to or during a separation by varying the applied potential. Stated differently, the conductive packing in EMLC can be uniquely viewed as a compositionally tunable stationary phase.
EMLC has been demonstrated for separating a wide range of charged analytes, including oligonucleotides and RNA [2]. Below, we discuss how it could be used to characterize various RNA QC attributes. The goal is not to claim that every existing QC assay can already be replaced, but to show that voltage-tunable conductive media may consolidate a meaningful subset of high-value separations now spread across several methods.
5' Capping Efficiency. It is the percentage of capped vs. uncapped RNA. The 5’ cap addition is critical for stability/translation, but also makes the RNA hydrophobic. Thus, reverse-phase HPLC is currently used to determine this attribute.
The All-Carbon reverse phase HPLC columns would separate capped and uncapped RNA species. EMLC may provide an additional tuning mechanism for cap-related separations beyond what a fixed reverse-phase method can offer. EMLC may offer useful flexibility for optimizing cap-related separations across different RNA constructs or process conditions without requiring new column chemistry each time.
Poly(A) tail presence and length. Poly(A) tail analysis is another area in which carbon-based EMLC appears particularly interesting. Poly(A) tails are highly hydrophobic and can adhere strongly to carbon surfaces, which creates a mechanistic basis for selective retention on all-carbon media.
This hydrophobicity opens up two related possibilities. First, EMLC may support conditions where poly(A)-containing species are selectively retained while other components are washed away. Then, these retained species can be released by changing the voltage. Second, gradually increasing the voltage during elution may achieve greater separation, thereby improving the apparent resolution of poly(A) tail-length distributions compared with a fixed-condition method.
Residual double-stranded RNA (dsRNA), solvent impurities, and aggregates. Residual double-stranded RNA is a high-priority impurity because of its immunogenicity. dsRNA differs from ssRNA in both structure and how it presents charge and hydrophobicity, and those differences may create useful separation behavior on carbon media.
An applied potential that pushes dsRNA away from the main ssRNA peak by altering electrostatic interactions at the carbon surface could be a useful starting point for method development. If that proves robust, EMLC could become a useful complement to conventional charge-based methods for impurity monitoring.
The same method logic could be extended to smaller process-related solvent impurities and to larger species such as aggregates. Small molecules and salts may separate early from the RNA peak. Larger aggregate species may respond differently to an electric potential than monomeric RNA, creating the possibility of a faster, purity-oriented screen that reduces exclusive dependence on a dedicated SEC workflow.
From separate methods to selective consolidation. The most compelling part of the RNA EMLC story is

not any single analyte. It is the possibility of selective consolidation. Today, QC laboratories often use HPLC systems with Column Switching Valves that can hold 4–6 different "monomodal" columns (e.g., SEC, RP, IEX columns). Theoretically, a user could switch from an RP LC column to an SEC LC column via software without moving the hardware. Thus, a single chassis can run them. However, standard QC labs rarely run these three methods on the same machine in a high-throughput environment. They always use separate, dedicated instruments due to solvent incompatibility, differences in hardware materials, and the time required to equilibrate between methods (changeover downtime). Collapsing these orthogonal dedicated workflows into a single LC separation would require a multimodal separation mechanism capable of interrogating all three physical properties simultaneously or sequentially within a single LC column.
Our long-term goal is not necessarily to eliminate every orthogonal assay, but to determine how far a single EMLC platform can go in covering multiple high-value RNA QC questions. A voltage-tunable all-carbon LC platform suggests a different model. Use a column-based architecture and modulate conditions to extract multiple classes of information with fewer handoffs between methods.
Summary and next steps. Even partial consolidation of RNA QC workflows could matter. Reducing the

number of dedicated methods saves time in equilibration, instrument allocation, and method maintenance — freeing capacity in QC labs that are already resource-constrained."
To that end, we are developing a bespoke EMLC system that combines custom electrically conductive porous carbon microbeads, 3D-printed monoliths, voltage-modulation electronics, potentiostat–LC column interfaces, and protocols specifically designed for RNA separations.
Our overarching goal is to determine how far a single voltage-tunable all-carbon platform can go in consolidating high-value RNA QC questions that today require multiple dedicated instruments.
For inquiries or collaborations, or if you are interested in evaluating NanoPak-C all-carbon microbeads, please contact our technical team at inquiry@millennialscientific.com, call us at 855 388 2800, or fill out our online contact form at www.millennialscientific.com/contact.
References
[1] J.A. Harnisch, M.D. Porter, Electrochemically Modulated Liquid Chromatography: An Electrochemical Strategy For Manipulating Chromatographic Retention, Analyst 126(11) (2001) 1841-1849.
[2] R. Lauman, H.J. Kim, L.K. Pino, A. Scacchetti, Y. Xie, F. Robison, S. Sidoli, R. Bonasio, B.A. Garcia, Expanding The Epitranscriptomic RNA Sequencing And Modification Mapping Mass Spectrometry Toolbox With Field Asymmetric Waveform Ion Mobility And Electrochemical Elution Liquid Chromatography, Analytical Chemistry 95(12) (2023) 5187-5195.
